Hologic Aquilex Fluid Management System Istruzioni per l'uso
- Tipo
- Istruzioni per l'uso

Form ENG-0034-T01, Rev. 005
TITLE
DOCUMENT NUMBER
REV
TEXT, IFU, AQUILEX FLUID
MANAGEMENT SYSTEM, 4320, NEW
SCALE
AW-16146-4320
004
SIZE A
SHEET 1 OF 1
REV AUTHORED BY
DATE
S.BHAMMAR
6/6/18
REV DRAFTED BY
DATE
N.FONSECA
12/16/20
PROPRIETARY: This document contains
proprietary data of Hologic, Inc. No
disclosure, reproduction or use of any part
thereof may be made except by written
permission from Hologic.
REV. RELEASE DATE:
1/12/2021
Artwork consists of:
• 302 8.26 inch x 11.69 inch (A4) sheets attached.
Artwork contains the following file(s):
File Name
Description
AW-16146-4320_004_02.pdf
View File
Artwork is black and white.

Instructions for Use en
Gebrauchsanweisung de
Instructions d’utilisation fr
Instrucciones de uso es
Istruzioni per l’uso it
Gebruiksaanwijzing nl

Manufacturer/Hersteller/Fabricant/Fabricante/Fabbricante/Fabrikant:
W.O.M. WORLD OF MEDICINE GmbH
Salzufer 8
10587 Berlin, Germany
Phone:+49 30 39981-550
Fax:+49 30 39981-545
E-mail:[email protected]
Distributed by/Vertrieb durch/Distribué par/Distribuido por/
Distributore/Distributeur:
HOLOGIC, INC.
250 Campus Drive, Marlborough
MA 01752 USA
1.800.442.9892 (US Toll Free)
1.508.263.2900 Type: H112 Bag scale / 1201601 / 10000021387 02
en Hologic and MyoSure are registered trademarks of Hologic,
Inc. and its subsidiaries in the United States and other coun-
tries. Aquilex is a trademark of Hologic, Inc. and its subsidiar-
ies in the United States and other countries. All other trade-
marks, registered trademarks, and product names are the
property of their respective owners.
These instructions for use contain information that is subject to copyright.
All rights reserved. These instructions for use may not be photocopied, du-
plicated on microfilm, or otherwise copied or distributed, completely or in
part, without the approval of W.O.M. WORLD OF MEDICINE GmbH.
We reserve the right to technical changes without prior notification due to
the continuous further development of our products. Function or design
may partially differ from the description in the instructions for use. Please
contact us for additional information about this or any of our other
products.
Some of the parts and equipment referred to in these instructions for use
are associated with registered trademarks but are not identified as such. It
should therefore not be assumed that the absence of the trademark symbol
indicates that any given designation is not subject to trademark protection.
Users of this product should not hesitate to point out to us any errors or is-
sues concerning these instructions for use.
Copyright © W.O.M. WORLD OF MEDICINE GmbH
de Hologic und MyoSure sind eingetragene Marken der Hologic,
Inc. und ihrer Tochtergesellschaften in den Vereinigten Staa-
ten und anderen Ländern. Aquilex ist eine Marke der Hologic,
Inc. und ihrer Tochtergesellschaften in den Vereinigten Staa-
ten und anderen Ländern. Alle anderen Marken, eingetrage-
nen Marken und Produktnamen sind Eigentum der jeweili-
gen Inhaber.
Diese Gebrauchsanweisung enthält eigentumsrechtlich geschützte Infor-
mationen, die dem Urheberrecht unterliegen. Alle Rechte vorbehalten. Oh-
ne ausdrückliche, schriftliche Genehmigung von W.O.M. WORLD OF MEDI-
CINE GmbH darf diese Gebrauchsanweisung weder vollständig noch in
Auszügen durch Fotokopie, Mikrofilm oder andere Verfahren vervielfältigt
oder verbreitet werden.
Durch die ständige Weiterentwicklung unserer Produkte behalten wir uns
technische Änderungen ohne Ankündigung vor. Funktion oder Design kön-
nen teilweise von der Beschreibung in der Gebrauchsanweisung abwei-
chen. Bitte kontaktieren Sie uns, um weitere Informationen zu diesem oder
anderen Produkten zu erhalten.
Bezeichnungen von Teilen und Zubehör, die zugleich eine eingetragene
Marke sind, wurden in dieser Gebrauchsanweisung nicht besonders ge-
kennzeichnet. Es kann nicht aus dem Fehlen des Markenzeichens geschlos-
sen werden, dass eine Bezeichnung keinen Markenschutz genießt.
W.O.M. WORLD OF MEDICINE GmbH ist Anwendern dieses Produkts dank-
bar für jeden Hinweis auf mögliche Fehler oder Unklarheiten dieser Ge-
brauchsanweisung.
Copyright © W.O.M. WORLD OF MEDICINE GmbH
fr Hologic et MyoSure sont des marques déposées de Hologic,
Inc. et de ses filiales aux États-Unis et d’autres pays. Aquilex
est une marque déposée de Hologic, Inc. et de ses filiales aux
États-Unis et d’autres pays. Toutes les autres marques,
marques déposées et désignations constituent la propriété
de leur détenteur respectif.
La présente instruction d'utilisation contient des informations protégées
par la législation des droits de propriété et des droits d’auteur. Tous droits
sont protégés. Il est interdit de reproduire ou de distribuer la présente ins-
truction d'utilisation - que ce soit intégralement ou partiellement par pho-
tocopie, micro-film ou autres procédés de reproduction sans l’autorisation
écrite expresse de l’entreprise W.O.M. WORLD OF MEDICINE GmbH.
En raison du perfectionnement permanent de nos produits, nous nous ré-
servons le droit de procéder à des modifications techniques sans avis préa-
lable. Il se peut que les fonctionnalités ou que le design des produits dif-
fèrent partiellement de la description figurant dans la présente instruction
d'utilisation. Pour de plus amples informations concernant ce produit ou
d’autres produits, prière de nous contacter.
Certains des éléments et équipements auxquels il est fait référence dans la
présente instruction d'utilisation sont associés à des marques déposées
mais pas spécifiquement identifiés comme tels. L'absence du logotype ne
peut en aucun cas laisser supposer que la désignation représente une
marque non déposée.
Les utilisateurs du présent produit sont vivement invités à signaler toute
erreur ou tout problème concernant la présente instruction d'utilisation.
Copyright © W.O.M. WORLD OF MEDICINE GmbH
es Hologic y MyoSure son marcas comerciales registradas de Ho-
logic, Inc. y sus subsidiarias en los Estados Unidos y otros paí-
ses. Aquilex es una marca comercial de Hologic, Inc. y sus sub-
sidiarias en los Estados Unidos y otros países. Todas las demás
marcas comerciales, marcas comerciales registradas y nom-
bres de productos son propiedad de sus respectivos dueños.
Estas instrucciones de uso contienen informaciones protegidas por el dere-
cho de propiedad (copyright), que forma parte de los derechos de autor. To-
dos los derechos están protegidos. Sin autorización de W.O.M. WORLD OF
MEDICINE GmbH, estas instrucciones de uso no podrá ser ni total ni parcial-
mente reproducidas ni divulgadas por medio de fotocopia, microfilm u
otros medios y procedimientos.
Debido al desarrollo constante de nuestros productos, nos reservamos el
derecho a llevar a cabo modificaciones técnicas sin aviso previo. El funcio-
namiento y el diseño podrán diferir parcialmente de la descripción en las
instrucciones de uso. Rogamos establezcan contacto con nosotros, si dese-
an adquirir más información sobre este o cualquier otro producto.
Algunas de las piezas y equipos a los que se hace referencia en estas ins-
trucciones de uso están asociados a marcas comerciales registradas pero
no se identifican como tales. La falta de la identificación con marca no im-
plica que el producto en cuestión no posea marca comercial alguna.
Los usuarios de este producto no deben dudar en señalarnos cualquier
error o cuestión relativa a estas instrucciones de uso.
Copyright © W.O.M. WORLD OF MEDICINE GmbH
Federal Law (only for U.S. market)
Caution: Federal law restricts this device to sale by or on the order of a physician.
Issued: 2020-10
MAN-05183-4320 Rev.003

it Hologic e MyoSure sono marchi registrati di Hologic, Inc. e re-
lative società affiliate negli Stati Uniti e in altri paesi. Aquilex
è un marchio di Hologic, Inc. e relative società affiliate negli
Stati Uniti e in altri paesi. Tutti gli altri marchi commerciali,
marchi registrati e nomi di prodotti sono di proprietà del ri-
spettivo proprietario.
Le presenti istruzioni per l’uso contengono informazioni soggette al diritto
d’autore. Sono riservati tutti i diritti. Senza espresso consenso scritto da
parte di W.O.M. WORLD OF MEDICINE GmbH non è consentito riprodurre
né pubblicare, per intero o parzialmente, le presenti istruzioni per l’uso me-
diante fotocopia, microfilm o altri procedimenti.
Grazie al continuo sviluppo dei nostri prodotti, ci riserviamo il diritto di ap-
portare modifiche tecniche senza alcun preavviso. Sia la funzione che il de-
sign possono scostarsi in parte dalla descrizione contenuta nelle presenti
istruzioni per l’uso. Vi preghiamo di volerci contattare per ulteriori informa-
zioni su questo o altri prodotti.
Le designazioni associate a marchi di fabbrica registrati non sono state par-
ticolarmente evidenziate. Dall’assenza di tale indicazione non si può dedur-
re che un’eventuale designazione rappresenti un marchio a libera disposi-
zione.
W.O.M. WORLD OF MEDICINE GmbH sarà grata a tutti gli utilizzatori dei
propri prodotti per qualsiasi indicazione su possibili errori o punti poco
chiari riscontrati nelle presenti istruzioni per l’uso.
Copyright © W.O.M. WORLD OF MEDICINE GmbH
nl Hologic en MyoSure zijn gedeponeerde handelsmerken van
Hologic, Inc. en haar dochtermaatschappijen in de Verenigde
Staten en andere landen. Aquilex is een handelsmerk van
Hologic, Inc. en haar dochtermaatschappijen in de Verenigde
Staten en andere landen. Alle andere handelsmerken, gede-
poneerde handelsmerken en productnamen zijn eigendom
van de desbetreffende houders.
Deze gebruiksaanwijzing bevat auteursrechtelijk beschermde informatie
waar copyright op bestaat. Alle rechten voorbehouden. Het is verboden om
deze gebruiksaanwijzing zonder toestemming van W.O.M. WORLD OF ME-
DICINE GmbH geheel of gedeeltelijk door middel van fotokopieën, micro-
film of met andere middelen te vermenigvuldigen of te verspreiden.
Door de voortdurende verdere ontwikkeling van onze producten behouden
wij ons het recht voor, zonder aankondiging vooraf technische wijzigingen
aan te brengen. De werking of het design kunnen in sommige gevallen af-
wijken van de beschrijving in het gebruiksaanwijzing. Neem voor meer in-
formatie over dit of andere producten contact met ons op.
Benamingen die tegelijkertijd een gedeponeerd handelsmerk zijn, zijn niet
speciaal gekenmerkt. Uit het ontbreken van het handelsmerk kan niet ge-
concludeerd worden dat het bij een benaming om een vrij handelsmerk
gaat. Evenmin kan hieruit worden afgeleid of er sprake is van octrooien of
gebruiksmodellen.
W.O.M. WORLD OF MEDICINE GmbH is gebruikers van W.O.M. WORLD OF
MEDICINE GmbH-producten dankbaar voor elke melding van mogelijke
fouten of onduidelijkheden in deze gebruiksaanwijzing.
Copyright © W.O.M. WORLD OF MEDICINE GmbH


Symbols
Symbols
Follow instructions for use
(white image on a blue back-
ground)
Consult instructions for use
Type BF applied part
Equipotentiality
Degrees of protection
provided by enclosures (IP-
Code)
Degrees of protection
provided by enclosures (IP-
Code)
Alternating current
Service
Catalogue number
Do not reuse
Do not resterilize
Sterilized using ethylene ox-
ide
Batch code
Serial number
Manufacturer
Date of manufacture (YYYY-
MM-DD)
Use by date (YYYY-MM-DD)
Quantity
Not made with phthalates
Made with phthalates
Keep dry
This way up
Fragile
Stacking limit by number
Keep away from sunlight
Protect from heat and radio-
active sources
Non-ionizing electomagnetic
radiation
Do not use if package is
damaged
en

Authorized for Sale or use by
Physician only
Temperature limit
Humidity limitation
Atmospheric pressure limita-
tion
Waste management
ON/OFF (push-push)
Max. use 30 days
Non sterile
Scale connection
Input/Output
Caution
Do not use this power out-
put
Certification Mark
Symbols
en

vii
Table of Contents
1 Important Operator/User Notes............................................................................................................................................................................9
2 Safety Information...................................................................................................................................................................................................10
3 Purpose........................................................................................................................................................................................................................11
3.1 Warnings and Precautions......................................................................................................................................................................... 11
3.1.1 Warnings .....................................................................................................................................................................................................11
3.1.2 Precautions .................................................................................................................................................................................................16
3.2 Description of the Aquilex® Fluid Control System.............................................................................................................................. 18
4 Initial System Set-Up ...............................................................................................................................................................................................19
4.1 Preparing the System for Use.................................................................................................................................................................... 19
4.2 System Components.................................................................................................................................................................................... 21
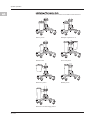
5 System Operation.....................................................................................................................................................................................................22
5.1 Front of Irrigation Pump Unit.................................................................................................................................................................... 22
5.2 Rear of Irrigation Pump Unit ..................................................................................................................................................................... 22
5.3 Fluid Monitoring Unit Set-up .................................................................................................................................................................... 23
5.3.1 Setting of the Container Scale ..............................................................................................................................................................26
5.3.2 Connecting the Vacuum Tube...............................................................................................................................................................27
5.4 Turning On the Aquilex® System ............................................................................................................................................................. 28
5.5 Hanging the Fluid Bags............................................................................................................................................................................... 29
5.6 Using Tube Sets............................................................................................................................................................................................. 30
5.7 Tube Overview............................................................................................................................................................................................... 30
5.8 Connecting the Outflow Tube Set ........................................................................................................................................................... 31
5.8.1 Connecting Outflow Tube of Tissue Removal Handpiece (e.g. MyoSure®)..............................................................................32
5.9 Inserting the Inflow Tube Set.................................................................................................................................................................... 33
5.10 Presetting the Intrauterine Pressure....................................................................................................................................................... 34
5.11 Deficit Limit Setting ..................................................................................................................................................................................... 35
5.12 Using the Pump during Surgery ............................................................................................................................................................... 35
5.13 Changing Bags during Surgery ................................................................................................................................................................. 36
5.14 Changing Container during Surgery ....................................................................................................................................................... 37
5.15 Changing Instrument during Surgery..................................................................................................................................................... 37
5.16 Total Inflow Volume Displayed................................................................................................................................................................. 38
5.17 Turning System Off ...................................................................................................................................................................................... 38
6 Function Test .............................................................................................................................................................................................................39
6.1 General Device and Setup Check.............................................................................................................................................................. 39
6.2 Scale Test......................................................................................................................................................................................................... 39
7 Safety Functions........................................................................................................................................................................................................40
8 Care and Maintenance............................................................................................................................................................................................41
8.1 Cleaning the System .................................................................................................................................................................................... 41
8.2 Maintenance Intervals ................................................................................................................................................................................ 41
8.3 Maintenance by Authorized Service Technician.................................................................................................................................. 41
8.4 Replacing of the Fuse................................................................................................................................................................................... 42
9 Annual Inspection.....................................................................................................................................................................................................43
9.1 Electrical Safety Test.................................................................................................................................................................................... 43
9.2 Basic Function Tests..................................................................................................................................................................................... 43
9.2.1 Scale Test.....................................................................................................................................................................................................43
9.2.2 Flow Rate Test............................................................................................................................................................................................44
9.2.3 Pressure Measuring Test.........................................................................................................................................................................45
9.2.4 Fluid Deficit Measurement Test ...........................................................................................................................................................46
9.2.5 Testing the Vacuum Pump.....................................................................................................................................................................47
9.3 Determine the Software Version ............................................................................................................................................................. 47
10 Error and Warning Messages................................................................................................................................................................................48
11 Technical Data...........................................................................................................................................................................................................49
12 Electromagnetic Compatibility.............................................................................................................................................................................51
12.1 Electrical Connections ................................................................................................................................................................................. 51
12.2 Guidance and Manufacturer's Declaration - Electromagnetic Emissions.................................................................................... 51
12.3 Guidelines and Manufacturer's Statement - Electromagnetic Interference Immunity........................................................... 52
en

viii
13 Accessory List.............................................................................................................................................................................................................54
14 Warranty Information.............................................................................................................................................................................................55
15 Appendix.....................................................................................................................................................................................................................56
15.1 Test log............................................................................................................................................................................................................. 56
Glossary.......................................................................................................................................................................................................................58
Index.............................................................................................................................................................................................................................59
en

Important Operator/User Notes
9 / 378
1 Important Operator/User Notes
Read the Instructions for Use carefully and become familiar with the operation and
function of the Aquilex® Fluid Control System (Aquilex® System) and the accessor-
ies before use during surgical procedures. Non-observance of the instructions listed
in this manual can lead to:
• life-threatening injuries to the patient,
• severe injuries of the surgical team, nursing staff or service personnel, or
• damage or malfunction of the system and/or accessories.
The Instructions for Use are only for the Aquilex® Fluid Control System, consisting
of pump, container scale and bag scale.
Subject to technical changes
The manufacturer reserves the right to modify the appearance, graphics, and tech-
nical data of the product through continued development of the product.
Please note
The words WARNING, CAUTION, and NOTE carry special meanings. Read these sec-
tions with special attention.
WARNING!
Warnings indicate risks to the safety of the patient or operator. Failure to follow
warnings may result in injury to the patient or operator.
CAUTION!
Warnings indicate risks to the equipment. Failure to follow cautions may result in
damage to the system.
NOTE!
These paragraphs contain information to clarify the instructions or provide addi-
tional useful information.
en

Safety Information
10 / 378
2 Safety Information
Exclusion of liability Hologic is not liable for indirect, incidental and consequential damages, including,
but not limited to, loss of profit. Any liability and applicable warranty become null
and void if:
• the system and/or the accessories/ peripherals are improperly used, transpor-
ted, stored, prepared, or maintained;
• the system and/or the accessories are improperly used, prepared, or main-
tained,
• the instructions and rules in the instructions for use are not adhered to;
• unauthorized persons perform repairs, adjustments, or alterations on the
device or accessories/peripherals;
• unauthorized persons open the device;
• the prescribed inspection and maintenance schedules are not adhered to.
Receipt of technical documentation from Hologic does not authorize individuals to
perform repairs, adjustments, or alterations on or to the system or accessories.
WARNING!Modification of the Aquilex® Fluid Control System is not per-
mitted.
Authorized service technician Only an authorized service technician may perform repairs, adjustments, or altera-
tions on the system or accessories/peripherals and use the service menu. Any viola-
tion will void any applicable warranty. Authorized service technicians are only
trained and certified by the manufacturer.
Normal Use The system may be used only as intended.
Care and maintenance The service and maintenance of the device and its accessories/ peripherals has to
be carried out as per instructions to ensure the safe operation of the device. For the
protection of the patient and the operating team, check that the device is complete
and functional before each use. Maintenance of the device may not be performed
during surgery or operation of the device.
NOTE!Service or maintenance work may not be carried out during surgery.
Contamination Before shipping, decontaminate the device and accessories/ peripherals in order to
protect the service personnel. Follow the instructions listed in these instructions for
use. If this is not possible,
• the product must be clearly marked with a contamination warning and
• is to be double-sealed in safety foil.
The manufacturer has the right to refuse the repair of contaminated devices or ac-
cessories/peripherals.
Waste management This symbol indicates that the waste of electrical and electronic equipment must
not be disposed as unsorted municipal waste and must be collected separately. For
disposal of the device and its accessories, please consult Hologic or an authorized
disposal company, in compliance with legal or national regulations.
en

Purpose
11 / 378
3 Purpose
Intended Use
The Aquilex® Fluid Control System is intended to provide fluid distension of the
uterus during diagnostic and operative hysteroscopies and to monitor the volume
differential between the irrigation fluid flowing into and out of the uterus.
Contraindications
The system may not be used to introduce fluids into the uterus when hysteroscopy
is contraindicated. See the operator’s manual of your hysteroscope for absolute
and relative contraindications.
Relative contraindications to endometrial ablation:
Hysteroscopic endometrial ablation, whether by laser or electrosurgery, should not
be undertaken before adequate training, preceptorship, and clinical experience.
Additionally, tissue sampling is required prior to destruction of the endometrium.
The following are clinical conditions that can significantly complicate hysteroscopic
endometrial ablation:
• Adenomatous endometrial hyperplasia
• Uterine leiomyoma
• Severe adenomyosis
• Pelvic pain (subtle PID)
• Uterine anomalies
• Surgical skill (see above)
• Severe anemia
• Inability to circumnavigate the myoma (re: myoma size) - predominantly intra-
mural myomas with small submucous components.
3.1 Warnings and Precautions
3.1.1 Warnings
WARNING!
Distension media
When performing monopolar hysteroscopic electrosurgery, the distension medium
must be electrically non-conductive. Examples include glycine, sorbitol and man-
nitol. Isotonic saline irrigation fluids may only be used when performing bipolar
electrosurgical resective procedures.
WARNING!
Pressure
The pressure should be kept as low as possible to allow for a sufficient intrauterine
distention and to reduce the forces that could allow fluid, ambient air, and/or gas
to enter the circulatory system.
WARNING!
Intrauterine distention
Intrauterine distention is usually possible with pressure values between 35 to
70mmHg. A pressure above 75mmHg is required only in rare cases or if the pa-
tient has an excessively high blood pressure.
WARNING!
Fluid overload
There is a risk of irrigation fluid reaching the circulatory system of the patient's soft
tissue by passing through the uterus. This can be affected by distention pressure,
flow rate, perforation of the uterine cavity and duration of the hysteroscopic sur-
gery. It is critical to closely monitor the input and outflow of the distending liquid
at all times.
en

Purpose
12 / 378
WARNING!
Fluid deficit
The fluid left in the patient must be monitored. The deficit is the total amount of
fluid left in the patient or unaccounted for otherwise. Take notice of the measure-
ment tolerance of the system (see ChapterTechnical Data [}49]). Estimating the
fluid volume remaining in the patient is the physician’s responsibility.
WARNING!
Fluid intake and output surveillance
Strict fluid intake and output surveillance should be maintained due to the risk of
fluid overload. For healthy patients, the maximum fluid deficit of 1,000ml is sug-
gested when using a hypotonic solution (e.g. glycine, sorbitol and mannitol). If iso-
tonic solutions (e.g. saline, Lactated Ringer's) are used, the fluid deficit should not
exceed 2,500ml.
WARNING!
Serum sodium concentration
It is also necessary to monitor the concentration of sodium in the blood of the pa-
tient to prevent electrolyte disturbances. Monitoring of the concentration of so-
dium in the blood must be performed by the physician and is not performed or sup-
ported by the system.
WARNING!
Loss of deficit and inflow value
The deficit display value is lost in case of a power loss or “brownout.”
WARNING!
Failure of scale connection
If the message “Check Scale Connection” appears, the deficit must be calculated
manually. The pump keeps displaying the last known deficit value determined prior
to the failure of the scale connection.
WARNING!
Container change during surgery
A container change during surgery is only allowed, if the container holds at least
0,5 liters of fluid. Otherwise, the deficit value may be falsified. In this case, the
manufacturer recommends manual deficit calculation.
WARNING!
Hyponatremia
Some distension fluids may lead to fluid overload and, consequently, hyponatremia
with its attending sequelae. This can be affected by the distending pressure, flow
rate, and duration of hysteroscopic procedure. It is critical to closely monitor the in-
put and outflow of the distending liquid at all times.
WARNING!
Pulmonary edema
Hysteroscopic surgery is associated with a risk of developing pulmonary edema res-
ulting from fluid overload with isotonic fluids. It is critical to closely monitor the in-
put and outflow of the distending liquid at all times.
en

Purpose
13 / 378
WARNING!
Cerebral edema
Hysteroscopic surgery is associated with a risk of developing cerebral edema result-
ing from fluid overload and electrolyte disturbances with hypoosmolar (non-ionic)
fluids such as glycine 1.5% and sorbitol 3.0%. It is critical to closely monitor the in-
put and outflow of the distending liquid at all times.
WARNING!
Idiosyncratic reactions
In rare cases, idiosyncratic reactions including
• intravascular coagulopathy
• allergic reaction including anaphylaxis
may occur while performing a hysteroscopy if a liquid distention medium is used.
WARNING!
Hypothermia (monitoring body temperature)
Continuous flow of distention fluids can lead to a lowering of the patient's body
temperature during hysteroscopic surgery. Lower body temperatures can cause
coronary and cardiovascular problems. Always monitor the patient's body temper-
ature during the entire surgery. Make especially sure that the following, hypother-
mia promoting, operation conditions are avoided as best as possible:
• longer operating times
• use of cold irrigation fluid.
WARNING!
Rupture of the fallopian tube secondary to tubal obstruction
Distention of the uterus may lead to a tear of the fallopian tube should there be an
obstruction or permanent occlusion. The rupture could lead to irrigation fluid flow-
ing into the patient's peritoneal cavity, resulting in a fluid overload. It is critical to
closely monitor the input and outflow of the distending liquid at all times.
WARNING!
Air embolism
An air embolism can be the result of air contained in the tube set or connected in-
strument reaching the patient. Ensure there is always fluid in the bag to prevent air
from being pumped into the patient.
WARNING!
Irrigation fluid bags
The system is only intended for use with flexible fluid bags. Do not use glass con-
tainers as they might break. With rigid containers, fluid cannot flow quickly
enough due to the vacuum being generated inside of the containers. Risk of implo-
sion with rigid containers.
WARNING!
Resetting the deficit display
Filling the tubing with irrigation fluid and resetting the deficit display to zero are to
be done at the physician’s discretion.
en

Purpose
14 / 378
WARNING!
Function test
The function test must be performed prior to each device use.
WARNING!
Obvious defects
Never use the system if it has suspected or confirmed defects, especially if these in-
volve the power plugs or the mains power supply connection cables. In this case
have the device repaired by authorized service personnel.
WARNING!
System error
Do not use the Aquilex® System if a defect is suspected or detected during the
function test. This also applies to any obvious defects, especially defects on the
power connector or plug and power cord.
WARNING!
Disconnect the power cord
Pressing the ON/OFF switch does not disconnect the system from the wall power
outlet. This requires pulling the power cord located in the rear of the system.
WARNING!
Technique and procedures
Only the physician can evaluate the clinical factors involved with each patient and
determine if the use of this device is indicated. The physician must determine the
specific technique and procedure that will accomplish the desired clinical effect.
WARNING!
Check all factory settings
Factory settings are not mandatory settings for the physician. The physician is re-
sponsible for all settings affecting the surgical procedure.
WARNING!
Original accessories
For your own safety and that of your patient, use only Aquilex® accessories (see
Chapter Accessory List [}54]).
WARNING!
Additional equipment
Additional equipment connected to medical electrical devices must be demon-
strated to be compliant with their respective IEC or ISO standards (IEC 60601-1,
IEC60950 or IEC62368 for data processing equipment). Furthermore, all configura-
tions must comply with the normative requirements for medical systems (see sec-
tion 16 of the last valid edition of IEC60601-1). Anyone who connects additional
devices to medical electrical equipment is a system configurator and as such is re-
sponsible for the system's compliance with the normative requirements for sys-
tems. Please contact the technical service if you have additional questions.
en

Purpose
15 / 378
WARNING!
Not explosion-proof
The system is not explosion-proof. Do not use in an area where flammable anes-
thetic gases are present.
WARNING!
Risk of electrical shock
To avoid risk of electrical shock, this system may only be connected to a supply
mains with protective earth.
WARNING!
Risk of electrical shock
To prevent electrical shock, do not open this device. Never open this device your-
self. Notify the authorized service technicians of any required repairs.
WARNING!
Professional qualification
The instructions for use do not include descriptions or instructions for surgical pro-
cedures/techniques. It is not suitable for training physicians in the use of surgical
techniques. Medical peripherals and devices may be used only by physicians or
medical assistants with the appropriate technical/medical qualifications working
under the direction and supervision of a physician.
WARNING!
Sterile media and accessories
Always work exclusively with sterile substances and media, sterile fluids, and
sterile accessories if so indicated.
WARNING!
Replacement device and accessories
In case the system or any of the accessories fail during a procedure, an alternative
system and replacement accessories should be kept within easy reach to be able to
finish the operation with the replacement components.
WARNING!
Condensation / Water penetration
Protect system from moisture. Do not use if moisture has penetrated the system.
WARNING!
Replace the fuse
Replace the fuse only with a fuse of the same type and rating (see Chapter Tech-
nical Data [}49]).
en

Purpose
16 / 378
WARNING!
Electromagnetic emissions
Electromagnetic emissions may increase and rise above the permissible limits if
other equipment (e.g. MyoSure® Control Unit) is stacked onto or placed directly
next to the Aquilex Fluid Control System. The user is responsible for monitoring the
devices to make sure they function properly.
WARNING!
If the Aquilex® Fluid Control System is configured as part of a ME SYSTEM, the en-
tire ME SYSTEM should be tested for compliance with IEC 60601-1-1, and any
equipment used with the Aquilex® Fluid Control System should be Type BF.
WARNING!
If the leakage current of the configured ME SYSTEM exceeds the limits of IEC
60601-1-1, install an appropriately rated UL 2601-1/IEC 60601-1 approved isolation
transformer and retest the system.
WARNING!
Incorrect determination of fluid deficit
Always use the hooks of the bag scale to hang the fluid bags to ensure an accurate
determination of the fluid deficit. In addition, leave the empty fluid bags hanging
on the bag scale until the end of surgery.
WARNING!
Filter
The vacuum tube with integrated filter is designed for max. 30 days. The vacuum
tube may not be sterilized. Replace the vacuum tube sooner if it is obviously con-
taminated. The filter prevents body fluids from entering the interior of the device.
3.1.2 Precautions
CAUTION!
Federal Law (only for U.S. market)
Federal law restricts this device to sale by or on the order of a physician.
CAUTION!
Indoor climate
Before switching on the device, sufficient time must have passed to adjust to the
indoor climate.
CAUTION!
Device setup
Device should be positioned outside of the sterile area in such a way that
• it can be easily disconnected,
• it is easy to use and switch off and on,
• it allows an easy monitoring of the display values, device functions, and access
to the control elements.
en

Purpose
17 / 378
CAUTION!
Modifying the system
Modifying the system and/or its accessories is not permitted.
CAUTION!
Combination of low set pressures and excessive vacuum pressures
When using the Aquilex® Fluid Control System with tissue removal systems, e.g.
MyoSure®, the combination of low set pressures and excessive vacuum pressures
may result in a significant loss of intrauterine distension pressure which has the
potential to affect the visibility of the surgical field. Conversely, when employing a
high distension pressure, the deactivation of the tissue removal system can lead to
pressure spikes that can exceed 150mmHg.
CAUTION!
Do not use the covered power output at the rear of the irrigation pump unit.
CAUTION!
Hysteroscope
The system may only be connected with hysteroscopes designed for and featuring
the technical specification permitting such a combined use. Any utilized hystero-
scopes must comply with the most recent versions of IEC60601-2-18 and ISO8600.
CAUTION!
Electrical interference
(see Chapter Electromagnetic Compatibility [} 51]): Electrical interference with
other devices or instruments was considered when developing this system and
none was detected during testing. However, if you still detect or suspect such inter-
ference, please follow these suggestions:
• Move the Aquilex® System, the other device, or both devices to a different loc-
ation.
• Increase distance between devices used.
• Consult an electro-medical expert.
CAUTION!
Wall outlet voltage
Check to ensure the available wall outlet voltage matches the data listed on the la-
bel attached to the back of the pump. Incorrect voltage can cause errors and mal-
functions and may destroy the system.
CAUTION!
Transport
The device is transportable. The roller wheels of the Fluid Monitoring Unit (cart/
scale) are used for positioning at the place of use. To transport the device, remove
all fluid bags from the hooks and make sure there are no containers or only com-
pletely emptied containers on the cart/scale. Inflow and outflow tubes must be
completely removed. Make sure the power supply line does not touch the ground
and there are no other objects located on the Aquilex® Fluid Control System. Al-
ways use the handle to move the system safely.
en

Purpose
18 / 378
CAUTION!
Accuracy of the deficit
To avoid affecting the accuracy of the deficit calculation ensure that the first step
of the canister change is to disconnect tubing from the full canisters. Reconnect
tubing to the new canisters only if they are already inserted into the scale.
CAUTION!
Combination of AQL-100PBS with AQL-100CBS
The irrigation pump unit AQL-100PBS may only be used with the fluid monitoring
unit AQL-100CBS as only this combination is approved to bear the MET NRTL mark.
CAUTION!
Cleaning the system / Sterilization not allowed
The pump and the cart/scale can be disinfected by wiping off the outer surfaces.
Do not sterilize the pump and the cart/scale.
3.2 Description of the Aquilex® Fluid Control System
Technical application scope of the
system
The intrauterine pressure can be adjusted on the front of the pump. It can be pre-
set to a range between 40 and 150mmHg. The maximum inflow rate is 800ml/
min and is reduced automatically by the pump once the pre-set intrauterine pres-
sure setting has been reached.
The system has been designed to provide both fluid and vacuum systems that max-
imize the performance of tissue removal systems, e.g. MyoSure®.
Suggested distension media The Aquilex® Fluid Control System can be used with hypotonic, electrolyte-free me-
dia (e.g., glycine 1.5% and sorbitol 3.0%) and isotonic, electrolyte containing media
(e.g., saline 0.9% and Lactated Ringer's).
Pressure measuring and regulating The system operates with a completely non-contact pressure measurement of the
irrigation medium. The contact-free pressure measurement is achieved by integrat-
ing the pressure chamber into the tubing system. The pressure chamber transmits
the irrigation fluid pressure to the electronics of the device via a pressure sensor.
The pressure control circuit continuously compares the desired preset intrauterine
pressure with the actual intrauterine pressure. The function of this algorithm is to
maintain the pre-set intrauterine pressure. Check for possible leaks if the pre-set
intrauterine pressure cannot be achieved.
en

Initial System Set-Up
19 / 378
4 Initial System Set-Up
Always check all parts and accessories of the system when performing initial setup.
If the system has obvious defects, contact Hologic Technical Support (Chapter War-
ranty Information [}55]).
Initial system set-up
Place the system on a level surface and install in a dry environment. The ambient
temperature and humidity must meet the requirements mentioned in Chapter
Technical Data [}49].
CAUTION!
Device setup
Device should be positioned outside of the sterile area in such a way that
• it can be easily disconnected,
• it is easy to use and switch off and on,
• it allows an easy monitoring of the display values, device functions, and access
to the control elements.
CAUTION!
Indoor climate
Before switching on the device, sufficient time must have passed to adjust to the
indoor climate.
4.1 Preparing the System for Use
CAUTION!
Possible malfunctions
The device Aquilex® Fluid Control System should not be used directly next to other
devices as this could result in malfunctions. The device Aquilex® Fluid Control Sys-
tem was tested for compliance with IEC60601-1-2 as a standalone system. There-
fore, do not stack other devices (e.g. MyoSure® Control Unit) on the system or the
Irrigation Pump Unit. In particular, do not place any other device than the
AQL-100PBS on the trays of the AQL-100CBS. If usage in the manner described
above is nevertheless required, this system and the other devices should be mon-
itored to make sure they function properly.
CAUTION!
Combination of AQL-100PBS with AQL-100CBS
The irrigation pump unit AQL-100PBS may only be used with the fluid monitoring
unit AQL-100CBS as only this combination is approved to bear the MET NRTL mark.
Mains connection
CAUTION!
Mains connection
• Make sure the available mains voltage matches the data listed on the type la-
bel attached to the back of the device. Incorrect voltage can cause errors and
malfunctions and may destroy the device.
• Make sure the connection data and technical specifications of the power sup-
ply comply with DIN VDE or national requirements. The mains connection
cable may be plugged only into a properly installed, grounded safety wall
socket (shockproof socket) (see DIN VDE 0100-710).
• Read the device label located in rear of device (type plate) to determine the op-
erating voltage of the device.
en
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
La pagina si sta caricando...
-
 1
1
-
 2
2
-
 3
3
-
 4
4
-
 5
5
-
 6
6
-
 7
7
-
 8
8
-
 9
9
-
 10
10
-
 11
11
-
 12
12
-
 13
13
-
 14
14
-
 15
15
-
 16
16
-
 17
17
-
 18
18
-
 19
19
-
 20
20
-
 21
21
-
 22
22
-
 23
23
-
 24
24
-
 25
25
-
 26
26
-
 27
27
-
 28
28
-
 29
29
-
 30
30
-
 31
31
-
 32
32
-
 33
33
-
 34
34
-
 35
35
-
 36
36
-
 37
37
-
 38
38
-
 39
39
-
 40
40
-
 41
41
-
 42
42
-
 43
43
-
 44
44
-
 45
45
-
 46
46
-
 47
47
-
 48
48
-
 49
49
-
 50
50
-
 51
51
-
 52
52
-
 53
53
-
 54
54
-
 55
55
-
 56
56
-
 57
57
-
 58
58
-
 59
59
-
 60
60
-
 61
61
-
 62
62
-
 63
63
-
 64
64
-
 65
65
-
 66
66
-
 67
67
-
 68
68
-
 69
69
-
 70
70
-
 71
71
-
 72
72
-
 73
73
-
 74
74
-
 75
75
-
 76
76
-
 77
77
-
 78
78
-
 79
79
-
 80
80
-
 81
81
-
 82
82
-
 83
83
-
 84
84
-
 85
85
-
 86
86
-
 87
87
-
 88
88
-
 89
89
-
 90
90
-
 91
91
-
 92
92
-
 93
93
-
 94
94
-
 95
95
-
 96
96
-
 97
97
-
 98
98
-
 99
99
-
 100
100
-
 101
101
-
 102
102
-
 103
103
-
 104
104
-
 105
105
-
 106
106
-
 107
107
-
 108
108
-
 109
109
-
 110
110
-
 111
111
-
 112
112
-
 113
113
-
 114
114
-
 115
115
-
 116
116
-
 117
117
-
 118
118
-
 119
119
-
 120
120
-
 121
121
-
 122
122
-
 123
123
-
 124
124
-
 125
125
-
 126
126
-
 127
127
-
 128
128
-
 129
129
-
 130
130
-
 131
131
-
 132
132
-
 133
133
-
 134
134
-
 135
135
-
 136
136
-
 137
137
-
 138
138
-
 139
139
-
 140
140
-
 141
141
-
 142
142
-
 143
143
-
 144
144
-
 145
145
-
 146
146
-
 147
147
-
 148
148
-
 149
149
-
 150
150
-
 151
151
-
 152
152
-
 153
153
-
 154
154
-
 155
155
-
 156
156
-
 157
157
-
 158
158
-
 159
159
-
 160
160
-
 161
161
-
 162
162
-
 163
163
-
 164
164
-
 165
165
-
 166
166
-
 167
167
-
 168
168
-
 169
169
-
 170
170
-
 171
171
-
 172
172
-
 173
173
-
 174
174
-
 175
175
-
 176
176
-
 177
177
-
 178
178
-
 179
179
-
 180
180
-
 181
181
-
 182
182
-
 183
183
-
 184
184
-
 185
185
-
 186
186
-
 187
187
-
 188
188
-
 189
189
-
 190
190
-
 191
191
-
 192
192
-
 193
193
-
 194
194
-
 195
195
-
 196
196
-
 197
197
-
 198
198
-
 199
199
-
 200
200
-
 201
201
-
 202
202
-
 203
203
-
 204
204
-
 205
205
-
 206
206
-
 207
207
-
 208
208
-
 209
209
-
 210
210
-
 211
211
-
 212
212
-
 213
213
-
 214
214
-
 215
215
-
 216
216
-
 217
217
-
 218
218
-
 219
219
-
 220
220
-
 221
221
-
 222
222
-
 223
223
-
 224
224
-
 225
225
-
 226
226
-
 227
227
-
 228
228
-
 229
229
-
 230
230
-
 231
231
-
 232
232
-
 233
233
-
 234
234
-
 235
235
-
 236
236
-
 237
237
-
 238
238
-
 239
239
-
 240
240
-
 241
241
-
 242
242
-
 243
243
-
 244
244
-
 245
245
-
 246
246
-
 247
247
-
 248
248
-
 249
249
-
 250
250
-
 251
251
-
 252
252
-
 253
253
-
 254
254
-
 255
255
-
 256
256
-
 257
257
-
 258
258
-
 259
259
-
 260
260
-
 261
261
-
 262
262
-
 263
263
-
 264
264
-
 265
265
-
 266
266
-
 267
267
-
 268
268
-
 269
269
-
 270
270
-
 271
271
-
 272
272
-
 273
273
-
 274
274
-
 275
275
-
 276
276
-
 277
277
-
 278
278
-
 279
279
-
 280
280
-
 281
281
-
 282
282
-
 283
283
-
 284
284
-
 285
285
-
 286
286
-
 287
287
-
 288
288
-
 289
289
-
 290
290
-
 291
291
-
 292
292
-
 293
293
-
 294
294
-
 295
295
-
 296
296
-
 297
297
-
 298
298
-
 299
299
-
 300
300
-
 301
301
-
 302
302
-
 303
303
-
 304
304
-
 305
305
-
 306
306
-
 307
307
-
 308
308
-
 309
309
-
 310
310
-
 311
311
-
 312
312
-
 313
313
-
 314
314
-
 315
315
-
 316
316
-
 317
317
-
 318
318
-
 319
319
-
 320
320
-
 321
321
-
 322
322
-
 323
323
-
 324
324
-
 325
325
-
 326
326
-
 327
327
-
 328
328
-
 329
329
-
 330
330
-
 331
331
-
 332
332
-
 333
333
-
 334
334
-
 335
335
-
 336
336
-
 337
337
-
 338
338
-
 339
339
-
 340
340
-
 341
341
-
 342
342
-
 343
343
-
 344
344
-
 345
345
-
 346
346
-
 347
347
-
 348
348
-
 349
349
-
 350
350
-
 351
351
-
 352
352
-
 353
353
-
 354
354
-
 355
355
-
 356
356
-
 357
357
-
 358
358
-
 359
359
-
 360
360
-
 361
361
-
 362
362
-
 363
363
-
 364
364
-
 365
365
-
 366
366
-
 367
367
-
 368
368
-
 369
369
-
 370
370
-
 371
371
-
 372
372
-
 373
373
-
 374
374
-
 375
375
-
 376
376
-
 377
377
-
 378
378
-
 379
379
Hologic Aquilex Fluid Management System Istruzioni per l'uso
- Tipo
- Istruzioni per l'uso
in altre lingue
Documenti correlati
-
 Hologic Aquilex Fluid Control System Istruzioni per l'uso
Hologic Aquilex Fluid Control System Istruzioni per l'uso
-
 Hologic NovaSure Istruzioni per l'uso
Hologic NovaSure Istruzioni per l'uso
-
 Hologic NovaSure Istruzioni per l'uso
Hologic NovaSure Istruzioni per l'uso
-
 Hologic NovaSure Istruzioni per l'uso
Hologic NovaSure Istruzioni per l'uso
-
 Hologic Eviva Stereotactic Guided Breast Biopsy System Istruzioni per l'uso
Hologic Eviva Stereotactic Guided Breast Biopsy System Istruzioni per l'uso
-
 Hologic ATEC Handpiece Istruzioni per l'uso
Hologic ATEC Handpiece Istruzioni per l'uso
-
 Hologic Brevera Breast Biopsy System Biopsy Needle Istruzioni per l'uso
Hologic Brevera Breast Biopsy System Biopsy Needle Istruzioni per l'uso
-
 Hologic ATEC Breast Biopsy and Excision System Introducer Localization System Istruzioni per l'uso
Hologic ATEC Breast Biopsy and Excision System Introducer Localization System Istruzioni per l'uso
-
 Hologic SecurMark Istruzioni per l'uso
Hologic SecurMark Istruzioni per l'uso
-
 Hologic SecurMark Istruzioni per l'uso
Hologic SecurMark Istruzioni per l'uso




































































































































































































































































































































































































